Publications
The Identity of Human Tissue-Emigrant CD8+ T Cells
Buggert, M., Vella,L.A., Nguyen, S., Wu, V., Sekine, T., Perez-Potti1, A., Maldini, C., Manne, S., Darko, S., Ransier, A., Kuri-Cervantes, L., Japp, A.S., Brody, I., Ivarsson, M., Hertwig, L., Antel, J.P., Johnson, M.E., Okoye, A, Picker, L., Vahedi, G., Sparrelid, E., Llewellyn-Lacey, S., Gostick, E, Björkström1, N., Bar-Or, A., Dori, Y., Naji, A., Canaday, D.H., Laufer, T.M, Wells, A., Price, D.A., Frank, I., Douek, D.C., Wherry, E.J, Itkin, M., Betts, M.R.
Cell
2020
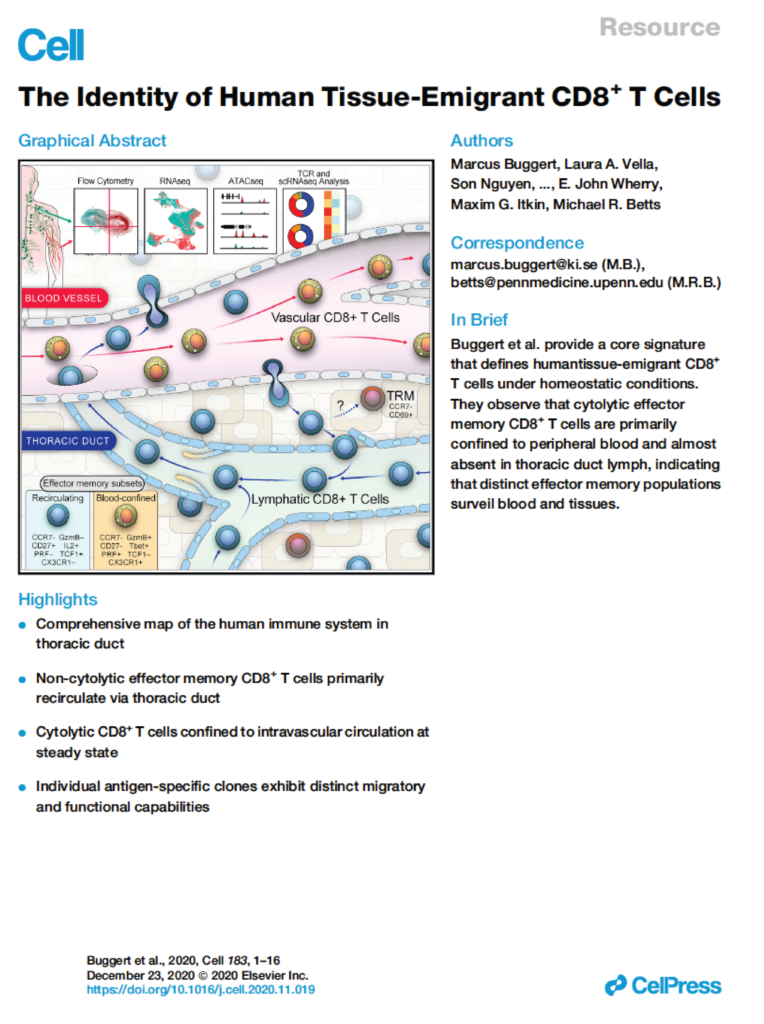
Lymphocyte migration is essential for adaptive immune surveillance. However, our current understanding of this process is rudimentary, because most human studies have been restricted to immunological analyses of blood and various tissues. To address this knowledge gap, we used an integrated approach to characterize tissue-emigrant lineages in thoracic duct lymph (TDL). The most prevalent immune cells in human and non-human primate efferent lymph were T cells. Cytolytic CD8+ T cell subsets with effector-like epigenetic and transcriptional signatures were clonotypically skewed and selectively confined to the intravascular circulation, whereas non-cytolytic CD8+ T cell subsets with stem-like epigenetic and transcriptional signatures predominated in tissues and TDL. Moreover, these anatomically distinct gene expression profiles were recapitulated within individual clonotypes, suggesting parallel differentiation programs independent of the expressed antigen receptor. Our collective dataset provides an atlas of the migratory immune system and defines the nature of tissue-emigrant CD8+ T cells that recirculate via TDL.